










| Current funding:
UArizona Health Sciences;
UArizona One Health Initiative;
KIOST
Past funding: Tucson Water; UArizona One Health Initiative; USDA/NIFA; UArizona Office of the Provost; Aqualung Therapeutics; TRIF; WET (NSF); Tucson Water; UArizona Test All Test Smart; KIOST; Tech Launch Arizona; CVTG (NIH); USDA/NIFA; BIO5 Institute; NSF/CBET; SWEHSC (NIH) Seoul VioSys; APQA; WSP; Desert Tech; WAESO (NSF); NVRQS; NIH/NIBIB; NSF/IUCRC; Arizona Department of Commerce; Theradiag; University of Arizona |
| Medical Diagnostics
Objective: Smartphone-based biosensors for diagnosing tissue, blood, saliva, and swab samples. Jocelyn Reynolds 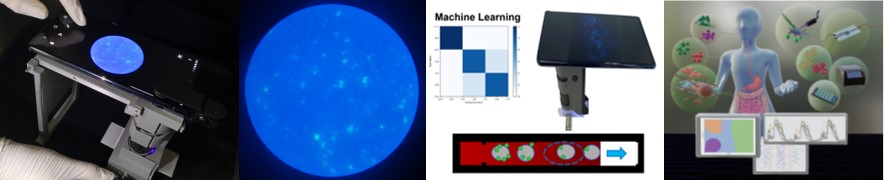
|
| 1-2. Smartphone fluorescence microscope takes images from a paper microfluidic chip for detecting COVID-19 from clinical samples.
3. NK cell subpopulation identification on a paper microfluidic chromatography using machine learning (ML).
4. Multispectral ML analysis of skin and gut microbiome. |
| Environmental Monitoring
Objective: Handheld biosensors for environmental monitoring. Lexi DeFord, Darya Pershina, Avery Miller, Janna Said, Benjamin Badalov 
|
| 1. Multidimensional ML analysis of capillary flow profiles for identifying bacterial species.
2. ML device identifies the oil type from oil spill samples.
3. Direct capture and in situ monitoring of viruses (COVID-19) from aerosols.
|
| Handheld RPA and PCR
Objective: Handheld RPA and PCR systems for virus and cancer identification. Reid Loeffler 
|
| 1. Emulsion LAMP is monitored with a smartphone for accurate and rapid detection of COVID-19.
2. The same can also be conducted on a Roche LightCycler for the detection of miRNA.
3. DOTS qPCR device with smartphone monitoring with under 5-min detection and inhibition relief. |
| Organ-on-a-Chip
Objective: Paper-based organ-on-a-chip for angiogenesis, drug/food toxicology, and blood-brain barrier studies. Trinity Hardy, Matthew Greenfield 
|
| 1-2. Paper-based liver-on-a-chip.
3. Angiogenesis on paper-based organ-on-a-chip.
4. Blood brain barrier (BBB) on a chip for toxicant and drug testing. |
Last revised: January 26, 2026.